In pharmaceutical manufacturing, the use of stainless steel piping in water purification systems has long been the standard. Recently, however, using polypropylene up to the point of distillation is challenging the status quo, and for good reason.
Let’s begin with some background on the safety standards within the pharmaceutical industry.
Regulations regarding piping material
Two governing bodies oversee the manufacturing of drugs. In the U.S., it is the United States Pharmacopoeia (USP) and in Europe, it is the European Pharmacopoeia (EP).
The guidelines issued by these two agencies are in accordance with strict regulations that are legally enforced by the Food and Drug Administration (FDA) and European equivalents like the Medicines and Healthcare Regulatory Agency (MHRA).
One of these regulations pertains to the piping material used from the point of the still forward.
Passivation can be costly to the bottom line
Both agencies require pharmaceutical manufacturers to use stainless steel piping after the point of distillation. The reason is simple: Stainless is able to withstand the intense heat generated during distillation.
Because stainless steel leads to rouging, the process of passivation becomes necessary.
The process of passivation restores the stainless steel piping to its original specifications by removing iron and debris from the surface and submerging the piping into a sanitizing bath.
Passivation is both expensive and time-consuming, taking a system down for 1-2 days, typically.
In the world of pharmaceutical manufacturing, this is a huge hit to the bottom line.
The Opportunity with Polypropylene
While Water for Injection (WFI) requirements make the use of stainless after the point of distillation necessary, there is opportunity for the use of polypropylene piping before the still.
In accordance with Water for Injection standards, when it comes to the distribution of pharmaceuticals in the United States and Europe, both the USP and the EP allow the use of polypropylene up to the point of distillation.
It’s an opportunity that some smart pharmaceutical manufacturers are starting to explore, and for good reason.
For API (Active Pharmaceutical Ingredients) plant facility applications, the use of polypropylene piping has huge benefits. Since the only downtime required with polypropylene is for routine disinfection, the cost advantages are significant.
This process can be done in hours, roughly once a year, versus the days required for passivation of stainless. Quite simply, repeated passivation is expensive.
Another benefit of polypropylene is that is produces the best quality of purified water. The integrity of the piping and the associated valves preclude the kind of microbial intrusion that may occur with stainless steel.
Components of a System with Polypropylene
Opaque Lines
When using polypropylene, it is critical to use opaque lines. Clear lines allow for light intrusion, that may lead to the growth of algae and biofilm.
Diaphragm Valves
With poly, the valves must be plastic diaphragm valves, not true union ball valves. Plastic diaphragm valves prevent corrosion and bacterial growth, while also ensuring optimal flow control.
IR Butt Fusion
In order to create a joint free of imperfections – imperfections that encourage the growth of bacteria – IR butt fusion must be employed. Diaphragm valves must be attached. Additionally, the machines used for IR butt fusion are designed to eliminate the possibility of human error, delivering a 99.9% success rate.
Six-Inch Pipe Diameter Dead-Leg System Requirement
With little to no flow in the pipe, there is a greater likelihood of microbial growth. Therefore, there must be no dead-leg piping more than six pipe lengths of the re-circulated piping loop, before the point-of-use batch tank.
Recapping Polypropylene – Turning Regulation into Opportunity
Pharmaceutical companies intending to distribute their products in both the United States and European countries must satisfy the requirements of the FDA and European equivalents like the MHRA.
One of these requirements is that any pharmaceutical company distributing product in the U.S. or Europe must use stainless steel piping from the point of distillation forward in their water purification system.
But when designed correctly, the integration of polypropylene piping up to the point of distillation is capable of ensuring product integrity, only for less money than water purification systems that exclusively use stainless steel.
For API facility applications, this difference provides a great cost advantage – one that will surely generate increased interest.
We’ll do more than give you a quote—we’ll visit your site to analyze your industrial water needs.

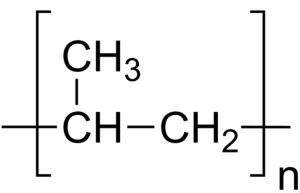
Follow Us